Steps 1 to 4 are repeated using. The mixture is stirred using a thermometer and the highest temperature reached is recorded.
Physical Amd Chemical Properties Of Ethanoic Aacid Chemistry Knowledgeuniverseonline Com
For example sodium hydroxide NaOH aq in its aqueous solutions dissociates as.

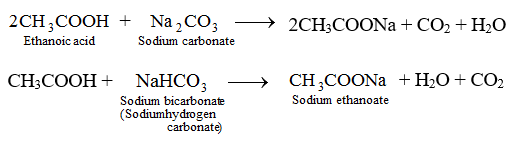
. The reaction between ethanoic acid and sodium carbonate. In other words the concentration of the ethanoate has to be half that of the ethanoic acid. NaOH HClO 4 NaClO 4 H 2 O.
ChlorineVII oxide itself also reacts with sodium hydroxide solution to give the same. ZnO H 2 SO 4 ZnSO 4 H 2 O. Hydrogen peroxide ortho-Phthaldehyde sodium hydroxide and sodium hypochlorite 70 phenol and 37 hydrochloric acid.
Sodium hydroxide Hydroxyde de sodium Caustic soda Lye Sodium hydrate. Oxygen nitrogen and the rare gases. The addition of hydroxide ions by adding lime sodium hydroxide or potassium hydroxide adjusts the pH because the hydroxide ion reacts with carbon dioxide to form bicarbonate alkalinity.
202297 1741 Mass percentage to Mass percentage. May be harmful if swallowed. US 01-145 bottle.
Some examples of bases are. Sodium iso-ascorbate Iso-ascorbate de sodium Sodium erythorbate Sodium salts of isoascorbic acid. Acetic acid ethanoic acid.
Our curricular based PL is offered to everyone. Ethanoic acid is the most common example of a carboxylic acid. Making Ethanoic Acid.
Acetone peroxide Peroxyde dacétone Peroxyacetone. There are elements for which spellings may differ such as aluminumaluminium sulfursulphur and. Sodium hydroxide solution is poured into a beaker of hydrochloric acid which contains a thermometer showing room temperature.
KOH CO 2 KHCO 3. High Quality Calcium Hydroxide Sale. H _ 2 SO_ 4.
One way of getting this for example would be to mix together 10 cm 3 of 10 mol dm-3 sodium ethanoate solution with 20 cm 3 of 10 mol dm-3 ethanoic acid. Harmful in contact with skin or if inhaled. These acids are a series of organic compounds that contain a carboxyl group COOH and there are a variety of methods that are used to produce them.
Strong Alkali Sodium Hydroxide with Best Price Min. The most common method is oxidising primary alcohols. NaCO 3 10H 2 O.
Identify two differences betweeh the two titration cur investigation. Ethanoic acid CH3COOH 36 1045 hydrochloric acid HCl 36 1180. Ammonia solution and ethanoic acid.
40 Tons Contact Now. The cards are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. Acetic acid Ethanoic acid.
To react with a base the amphoteric hydroxide often needs to have been freshly produced and the base must be hot and concentrated. Students should be able to. Ammonia solution and hydrochloric acid.
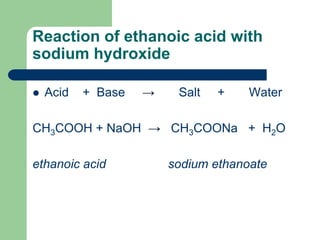
DEthene is burnt in presence of oxygen to form carbon dioxide water and releases heat and light. Predict the charge on a simple ion using the position of the element in the Periodic Table. BSodium hydroxide solution is treated with acetic acid to form sodium acetate and water.
Hydrated sodium carbonate. The mass of calcium carbonate in an indigestion tablet. Propanone Acetone Propene Propylene Sodium carbonate.
Graph the pH versus NaOH added. NaOH aq Na aq OH aq The term alkali is often used for water soluble bases. Flammable liquid and vapor.
Sodium hydroxide solution and ethanoic acid. This is a list of common chemical compounds with chemical formulae and CAS numbers indexed by formulaThis complements alternative listing at list of inorganic compoundsThere is no complete list of chemical compounds since by nature the list would be infinite. A base is a substance which furnishes hydroxide ions OH when dissolved in water.
202297 1741 Volume-volume percentage to Normality. The ICSC project is a common undertaking between the World. I Sodium hydroxide NaOH or caustic soda used in washing soaps.
The concentration of ethanoic acid in vinegar. The formulas of compound ions eg sulfate hydroxide nitrate carbonate and ammonium. The primary aim of the cards is to promote the safe use of chemicals in the workplace.
Identify any buffer regions on your titration curve. Because these molecules do not fully dissociate the pH shifts less when near the equivalence point. Beyond the equivalence point when the sodium hydroxide is in excess the curve is just the same as that end of the HCl - NaOH graph.
Acetone acetonitrile ACN benzene ethanol diethyl ether isopropanol methanol methyl ethyl ketone tetrahydrofuran THF. Phosphorous acid has a pK a of 200 which makes it stronger than common organic acids like ethanoic acid pK a 476. The hydrochloric acid is then poured quickly and carefully into the sodium hydroxide solution.
Ethanoic acid Acetic acid Ethanol. Titration curves for weak acid v weak base. Draw and fully label a titration curve from the data collected from the acetic ethanoic acid and sodium hydroxide titration.
Page 6 of 11. CEthanol is warmed with ethanoic acid to form ethyl acetate in presence of cone. Health Hazards of KOH.
Sodium hydroxide NaOH 10 1110. SSERC offers a vast portfolio of professional learning PL programmes for STEM educators in Scotland. Acetic anhydride Anhydride acétique Acetic oxide Acetyl oxide Ethanoic anhydride.
MgSO 4 7H 2 O. The start of the graph shows a relatively rapid rise in pH but this slows down as a buffer solution containing ethanoic acid and sodium ethanoate is produced. In the following example zinc oxide becomes the zincate ion ZnOH 4 as part of soluble sodium zincate when added to concentrated base.
The main target users are workers and those responsible for occupational safety and health. Factory Laboratory CAS 64-19-7 Ethanoic Acid Chemical Reagent Acetic Acid FOB Price. Or 10 cm 3 of 10 mol dm-3 sodium ethanoate solution with 10 cm 3 of 20 mol dm-3 ethanoic acid.
This is done in two simplified stages. Acetic acid ə ˈ s iː t ɪ k systematically named ethanoic acid ˌ ɛ θ ə ˈ n oʊ ɪ k is an acidic colourless liquid and organic compound with the chemical formula CH 3 COOH also written as CH 3 CO 2 H C 2 H 4 O 2 or HC 2 H 3 O 2. Childminders early years workers primary and secondary staff as well as lecturers technicians and those who work with young people in non-formal settings such as youth workers and in the CLD sector.
Mass-volume percentage to Molarity Recent user inquiry. 125 dihydroxycholecalciferol 1-Day 1-deamino-8-d-arginine vasopressin 13-cis-retinoic acid 2-deoxycoformycin 2-amino-6-mercaptopurine 2-amino-6-trifluoromethoxy-benzothiazole 2-CdA 2-chlorodeoxyadenosine 2-PAM 2-propylpentanoic acid 2-propylvaleric acid 2-pyridine aldoxime methochloride 292 MEP 311C90 3M Avagard Discontinued 3M Cavilon Skin Cleanser 3TC 4. ChloricVII acid reacts with sodium hydroxide solution to form a solution of sodium chlorateVII.
An example of a weak acid is acetic acid ethanoic acid and an example of a weak base is ammonia. The health hazards of potassium hydroxide are similar to those of the other strong alkalies such as sodium. 100 bottle Contact Now.
Vinegar is at least 4 acetic acid by volume making acetic acid the main component of vinegar apart from water and other trace elements. Caustic Soda Price Caustic Soda Manufacturer Caustic Soda for Soap.

How To Balance Naoh Ch3cooh Ch3coona H2o Youtube

List Two Chemical Properties On Basis Of Which Ethanol Ethanoic Acid

Write Chemical Equations For What Happens When Sodium Metal Is Added To Ethanoic Acid Solid Sodium Carbonate Is Added To Ethanoic Acid Ethanoic Acid Reacts With A Dilute Solution Of
0 Comments